For quotations, please use our online quotation form, and you may also contact us by
sales@neoscientific.com
+1-888.733.6849
+1-617.299.7367 (Int’l)
+1-888.733.6849
+1-617.299.7367 (Int’l)
| Reactivity | Human Mouse Rat |
| Tested applications | WB IHC IF |
| Recommended Dilution | WB 1:500 - 1:2000 IHC 1:50 - 1:100 IF 1:10 - 1:100 |
| Calculated MW | 77kDa |
| Observed MW | Refer to Figures |
| Immunogen | A synthetic peptide of human PRKCA |
| Storage Buffer | Store at -20℃. Avoid freeze / thaw cycles. Buffer: PBS with 0.02% sodium azide, 50% glycerol, pH7.3. |
| Concentration | bqr |
| Synonym | PKCA;AAG6;PKCA;PRKACA; PKC-alpha; MGC129900; MGC129901; PRKCA |
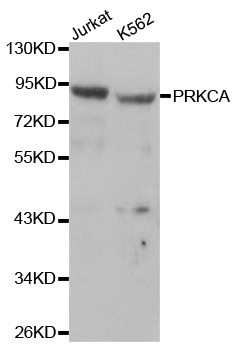
Western blot analysis of extracts of various cells, using PRKCA antibody.
Immunohistochemistry of paraffin-embedded human lung cancer using PRKCA antibody at dilution of 1:100 (40x lens).

Immunohistochemistry of paraffin-embedded human colon using PRKCA antibody at dilution of 1:100 (40x lens).

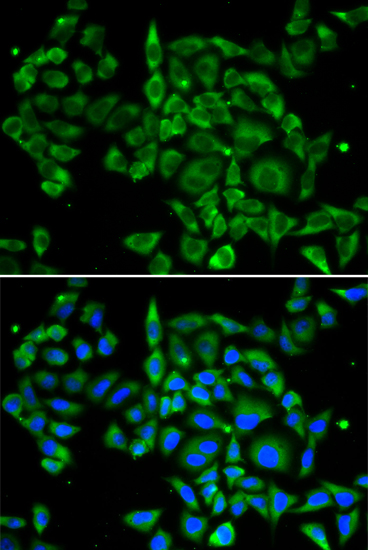
Immunofluorescence analysis of U2OS cell using PRKCA antibody. Blue: DAPI for nuclear staining.
Activation of protein kinase C (PKC) is one of the earliest events in a cascade that controls a variety of cellular responses, including secretion, gene expression, proliferation, and muscle contraction (1,2). PKC isoforms belong to three groups based on calcium dependency and activators. Classical PKCs are calcium-dependent via their C2 domains and are activated by phosphatidylserine (PS), diacylglycerol (DAG), and phorbol esters (TPA, PMA) through their cysteine-rich C1 domains. Both novel and atypical PKCs are calcium-independent, but only novel PKCs are activated by PS, DAG, and phorbol esters (3-5). Members of these three PKC groups contain a pseudo-substrate or autoinhibitory domain that binds to substrate-binding sites in the catalytic domain to prevent activation in the absence of cofactors or activators. Control of PKC activity is regulated through three distinct phosphorylation events. Phosphorylation at Thr500 in the activation loop, the autophosphorylation site at Thr641, and at carboxy-terminal hydrophobic site Ser660 occurs in vivo (2). Atypical PKC isoforms lack hydrophobic region phosphorylation, which correlates with the presence of glutamic acid rather than the serine or threonine residues found in more typical PKC isoforms. Either the enzyme PDK1 or a close relative is responsible for PKC activation. A recent addition to the PKC superfamily is PKCμ (PKD), which is regulated by DAG and TPA through its C1 domain. PKD is distinguished by the presence of a PH domain and by its unique substrate recognition and Golgi localization (6). PKC-related kinases (PRK) lack the C1 domain and do not respond to DAG or phorbol esters. Phosphatidylinositol lipids activate PRKs and small Rho-family GTPases bind to the homology region 1 (HR1) to regulate PRK kinase activity (7).
N/A