For quotations, please use our online quotation form, and you may also contact us by
sales@neoscientific.com
+1-888.733.6849
+1-617.299.7367 (Int’l)
+1-888.733.6849
+1-617.299.7367 (Int’l)
INTRODUCTION
NeoFectin® Transfection Reagent is a non-Liposomal, chemically-defined, high-performance, broad spectrum transfection reagent that provides highly efficient transfection of DNA/RNA into mammalian cells, including hard-to-transfect cell types, such as primary cells and stem cells. It is serum compatible, low in toxicity, and easy to use.

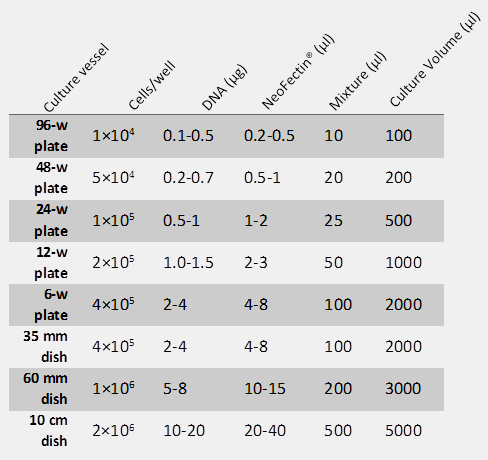
General recommendations for maximum efficiency
Nucleic acids: Highly purified, endotoxin-free and contaminant-free nucleic acids are recommended.
Cell density: We recommend splitting cells 18-24 hours prior to transfection. Cells should be 40-80% confluent at the time of transfection.
Ratio of NeoFectin® to nucleic acid: We recommend using 2µl of NeoFectin® per microgram of nucleic acids. This 2:1 ratio may be varied from 1:1 to 1:4 (µl NeoFectin®: µg nucleic acid) until optimal efficiency is achieved. Recommended starting conditions table provides ratio based on cell culture vessel size.
TRANSFECTION PROTOCOL
The following protocol is for transfection of adherent cells in a 6-well plate. For different plate formats follow the recommendations in the Table.
18–24 hours before transfection split cells to achieve 70% confluence at the time of transfection. This equates to plating approximately 4×105cells/well. Each well should contain 2ml of complete growth medium.
Add 2 µl DNA/or RNA (1 µg/µl) to 94µl complete growth medium.
Add 4µl NeoFectin® and vortex gently.
Incubate at room temperature 15-20 minutes.
Use a pipette to evenly distribute the mixture to the well. There is no need to change medium before adding transfection mixture.
Rock the plate to fully distribute the Neofectin®/nucleic acid complexes.
Incubate at 37°C, 5% CO2.
There is no need to change the medium after adding transfection mixture. If desired medium can be changed 4-6 hours after adding complexes without significant effects on efficiency.
Uptake of NeoFectin®/nucleic acid complexes will continue for several hours. If a highly active expression vector is used, expression should be evident within 4-6 hours.
Non-adherent cells can also be transfected. We recommend using the protocol described here but starting with 8×105 cells in 2ml of complete medium. Scale based on ratios given here.
To generate stable cell lines begin selection 24-48 hours after transfection.